Lunit AI Applied to Clinical Trial for Drug Development for the First Time--Findings Presented at ESMO 2021
Sep 17, 2021 - Lunit MediaLunit SCOPE, an AI-based tissue analysis platform, has been applied to a clinical trial of a new drug for the first time, accurately predicting immunotherapy response of the patients.
Two products of Lunit SCOPE--Lunit SCOPE IO and Lunit SCOPE PD-L1--ready to be launched at the end of this year.
A leading medical AI provider Lunit today announced that its AI for tissue analysis has been applied in a clinical trial for drug development, accurately predicting the patients’ response to immunotherapy. Including this major finding, the company presented three studies at European Society for Medical Oncology(ESMO) Congress 2021.
Lunit has been focusing on developing novel AI biomarkers that can be applied in cancer treatment. Since 2019, it has been validating the effectiveness of its AI-based tissue analysis platform ‘Lunit SCOPE’ that accurately predicts cancer patients’ response to immunotherapy.
The highlight of Lunit at ESMO 2021 is its announcement that Lunit SCOPE IO—one of Lunit SCOPE product lines—has been applied to a phase 1 clinical trial of a new drug for the first time. In a joint study with Y Biologics, a South Korean clinical-stage antibody biotech company, Lunit SCOPE IO accurately predicted immunotherapy response of all the patients who participated in the study.
“From 2019, Lunit has proven that response to immunotherapy varies depending on the spatial distribution of immune cells in cancer tissue, which has been the foundation for the AI platform ‘Lunit SCOPE IO’,” said Chan-Young Ock, Chief Medical Officer of Lunit. “This study is the first case where Lunit SCOPE IO was applied to actual drug development clinical trials, and it is a meaningful study that demonstrated high potential for AI-powered biomarkers.”
Lunit presented another study using Lunit SCOPE IO, validating the correlation between spatial information of tumor-infiltrating lymphocytes(TIL) and prognosis of colorectal cancer. The team analyzed 461 colorectal cancer data among TCGA data and showed that high TIL density in relation to tumor cells may improve prognostic power.
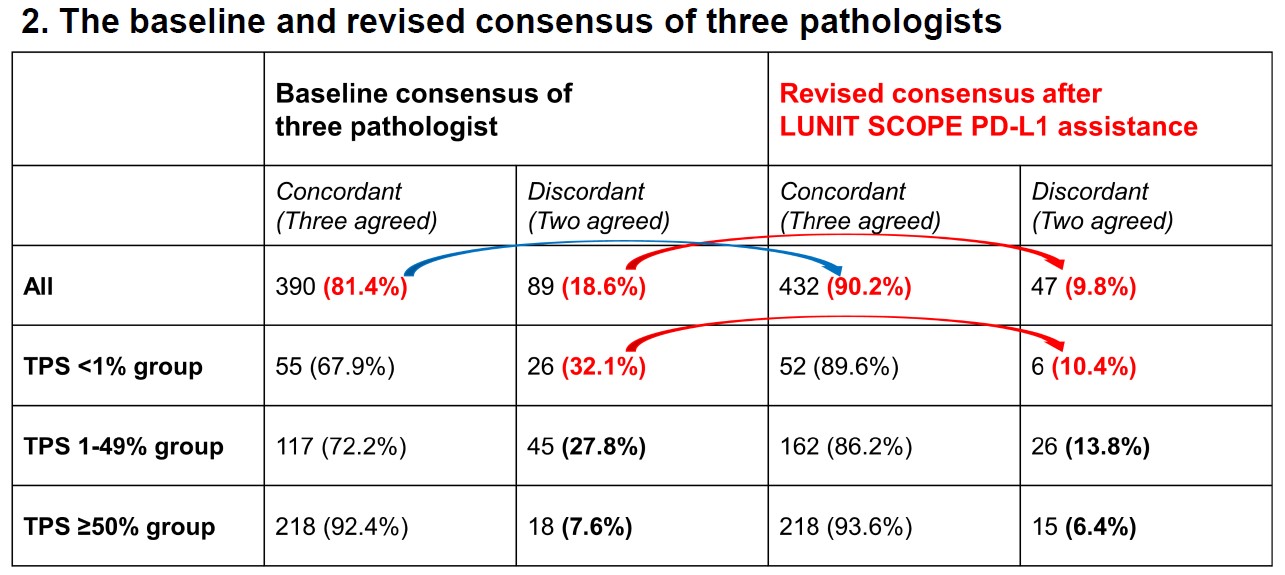
The company’s other major study was on Lunit SCOPE PD-L1, an AI-powered PD-L1 tumor proportion score(TPS) analyzer developed by Lunit. According to the study, assistance with Lunit SCOPE PD-L1 substantially improved pathologists’ consensus and found more patients who were eligible for immunotherapy.
“Although PD-L1 expression is the standard biomarker for advanced non-small cell lung cancer, manual evaluation of PD-L1 TPS by pathologists has practical limitations of interobserver bias and intensive labor,” said Kyunghyun Paeng, Chief Product Officer of Lunit. “This study aimed to explore whether the AI-powered TPS analyzer could reduce the interobserver variation and increase the accuracy of analysis.”
Three pathologists evaluated the PD-L1 TPS of 479 NSCLC data. Comparing the results of the analysis, the discordant rate in the subgroups of TPS <1% decreased from 32% to 10% when assisted with Lunit AI. Furthermore, 23 out of 81 were found to be eligible for immunotherapy even though they were evaluated to be TPS <1% by pathologists, meaning an additional 30% of patients could be treated with immunotherapy, who would otherwise have not been recommended for immunotherapy.
“We are now in the process of proving and creating the direction we want to achieve in the field of cancer treatment,” said Brandon Suh, CEO of Lunit. “This year’s ESMO is particularly meaningful as one of our Lunit SCOPE products was used in a clinical trial for the first time and showed its predictable value. Our validation of AI-powered biomarkers for immunotherapy will be applied to many other drugs and larger clinical studies in the near future. In light of these positive results, Lunit SCOPE will be commercially made available by the end of this year.”
Lunit Abstract Information at ESMO 2021
Virtual Meeting: E-Posters
September 16 - 21, 2021
Title: Interim results of phase I dose escalation study of YBL-006: A novel anti-PD-1 monoclonal antibody in advanced solid tumors
Title: AI-powered whole-slide image analysis of tumor-infiltrating lymphocytes for prediction of prognosis in colorectal cancer
Title: Assistance with an artificial intelligence-powered PD-L1 analyzer reduces interobserver variation in pathologic reading of tumor proportion score in non-small cell lung cancer