-
Lunit AI products for analyzing chest X-ray and mammograms now approved to support medical professionals ofSingapore, by detecting suspicious lesions with 96-99% accuracy
-
Applied in one ofSingapore'sleading healthcare centers, with positive feedback from the users
Lunit today announced its AI products for analyzing chest X-ray 'Lunit INSIGHT CXR' and breast mammography 'Lunit INSIGHT MMG' have received HSA approval to be commercially available inSingapore.
As a leading medical AI company, Lunit provides AI software that detects and analyzes radiology images. The most mature products--Lunit INSIGHT CXR and Lunit INSIGHT MMG--provide AI-powered detection of lesions that are suspicious of chest abnormalities and breast cancer with 96-99% accuracy and visualizes the location of the findings.
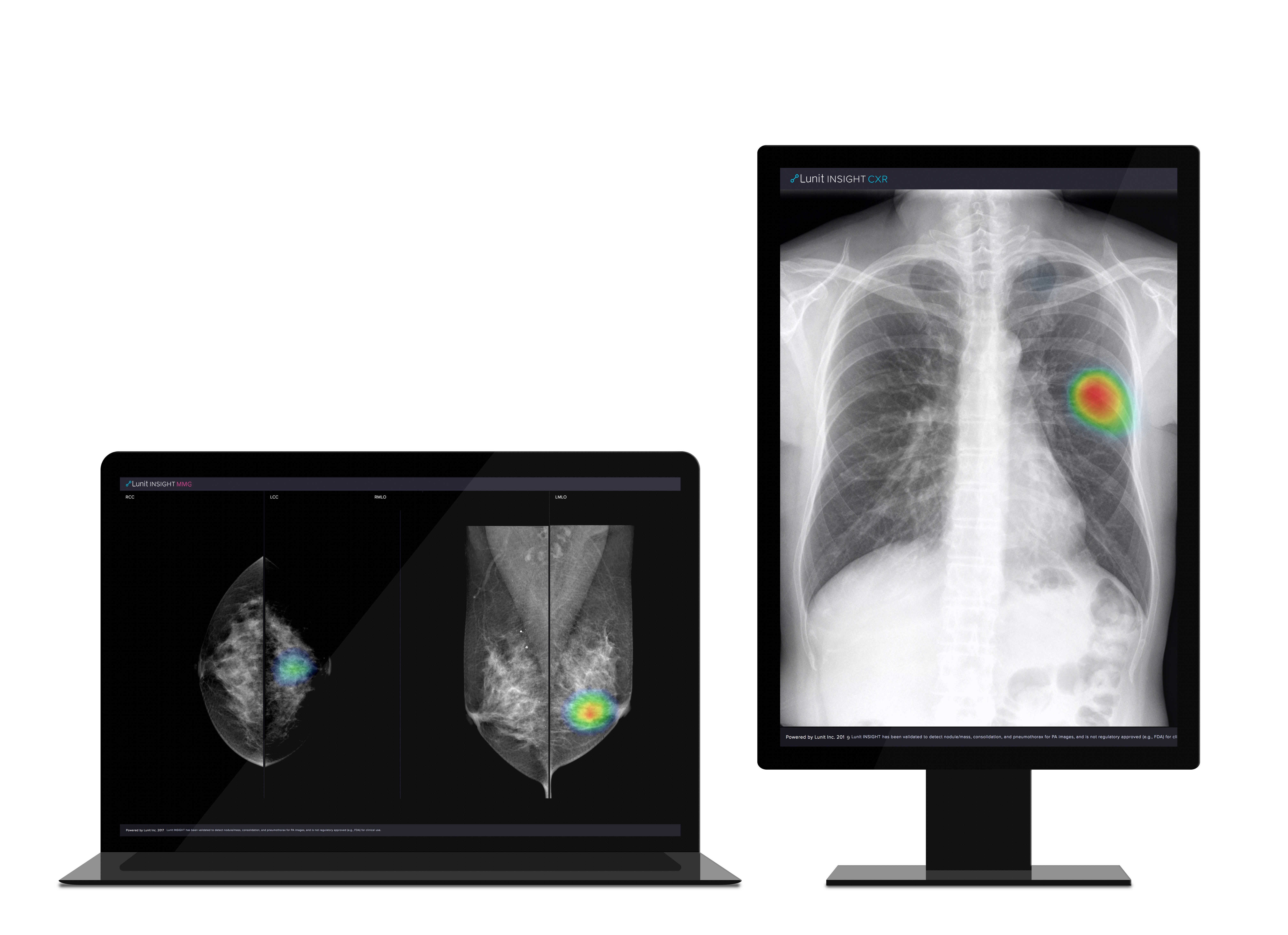
Image of Lunit INSIGHT MMG (right) and Lunit INSIGHT CXR (left)
"Singaporeis a healthcare hub of APAC, known to provide high quality and easy access to medical service for patients," saidBrandon Suh, CEO of Lunit. "We are delighted to introduce our AI solution to hospitals and medical centers inSingapore, to create clinical value and ensure healthier life for the people."
As for its first step, the company's AI solution has been applied to FeM Surgery, one ofSingapore'sleading healthcare centers holding eight different centers aroundAsia. After validating through a research model, the center uses AI in clinical practice for breast cancer screening.
"Lunit AI has proven to be a reliable second reader in my practice," said Dr.Felicia Tan, Director of FeM Surgery. "It has sped up the daily workflow and at the same time improved accuracy in mammogram interpretation."
Lunit INSIGHT products are now clinically used in over 300 sites worldwide to help medical professionals more efficiently and accurately diagnose diseases in chest X-rays and mammography images. It is CE marked and also holds approval in more than 30 countries including Korea,Japan,Indonesia,Australia,Brazil,Mexico,South Africa, and more.